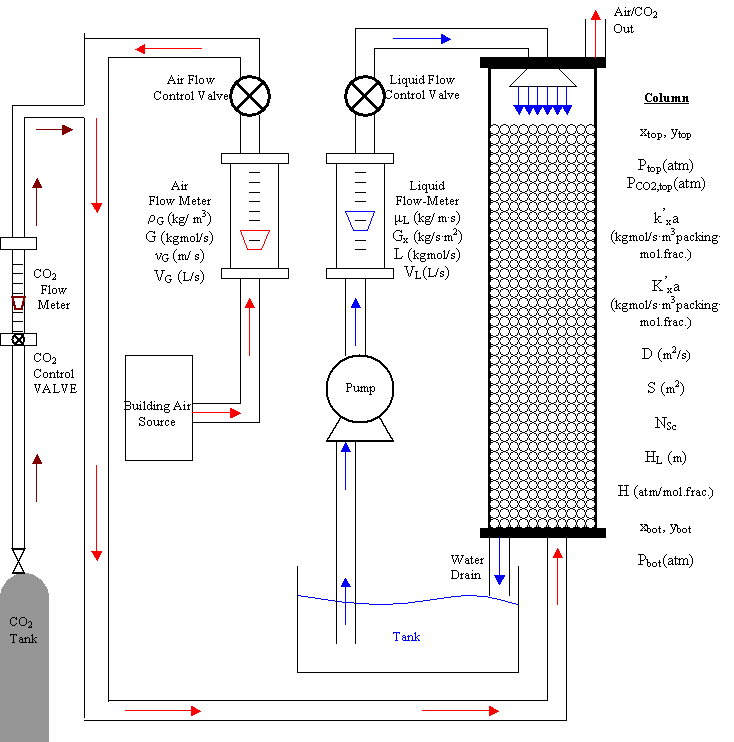

The variables listed on the process flow diagram above will be encountered when performing the experimental and theoretical calculations for Experiment 380. Table I below defines the symbols and provides the appropriate units.
Table I: Pertinent Symbols and Definitions
|
Variable |
Name |
Units |
|---|---|---|
|
rg |
gas density |
kg/ m3 |
|
rL |
liquid density |
kg/ m3 |
|
D |
Diffusivity |
m2/s |
|
G |
gas flow rate |
kgmol/s |
|
Gx |
gas mass velocity
per cross sectional area |
kg/s×m2 |
|
H |
Henry’s Law
constant |
atm/mol.frac. |
|
HL |
Liquid film height |
m |
|
k’xa |
liquid film mass transfer coefficient |
kgmol/s·m3packing·mol.frac |
|
K’xa |
overall mass
transfer coefficient |
kgmol/s·m3packing·mol.frac |
|
L |
liquid flow rate |
kgmol/s |
|
NSc |
Schmidt number |
- |
|
Pbot |
total pressure at
bottom of column |
atm |
|
PCO2,bot |
CO2
pressure at bottom of column |
atm |
|
PCO2,top |
CO2
pressure at top of column |
atm |
|
Ptop |
total pressure at
top of column |
atm |
|
S |
cross sectional
area |
m2 |
|
nG |
Gas Velocity |
m/s |
|
VG |
volumetric flow
rate of gas |
L/min |
|
VL |
volumetric flow
rate of liq |
L/min |
|
x*bot |
CO2
bottom mole frac. at equilibrium |
- |
|
x*top |
CO2 top
mole frac. at equilibrium |
- |
|
xbot |
liquid phase CO2
mole frac. at the bottom of the column |
- |
|
xtop |
liquid phase CO2
mole frac. at the top of the column |
- |
|
ybot |
Gas phase CO2
mole frac. at the bottom of the column |
- |
|
ytop |
gas phase CO2
mole frac. at the top of the column |
- |
|
μL |
liquid viscosity |
kg/mּs |
Description
of Variables
rg is the gas density. It is used to determine the gas flow rate in terms of mass per second.
rL is the liquid density. It is used to determine the liquid flow rate in terms of mass per second. It is also used to determine the Schmidt number.
D is the diffusivity of a gas solute into a liquid solvent. It is used to determine the Schmidt number.
G is the gas flow rate in terms of mass of total gas. It is used in the mass balance to calculate the composition of gas or liquid streams.
Gx is the gas flow rate by mass (G) divided by the cross sectional area of the column (S). It is used to calculate the empirical value of the mass transfer coefficient.
H is the Henry’s law constant. It is used to calculate the equilibrium concentration of solute in a liquid stream given a known gas composition and pressure at that same point.
HL is the liquid film height. It is used to calculate an empirical value for the mass transfer coefficient.
k’xa is the liquid side film mass transfer coefficient. It is the proportionality constant that describes the mass transfer that occurs on the liquid side of an interface.
K’xa is the overall mass transfer coefficient. It is a combination of the liquid and gas film mass transfer coefficient. It is the proportionality constant for the overall mass transfer in the system.
L is the liquid flow rate in terms of mass of total gas. It is used in the mass balance to calculate the composition of gas or liquid streams.
NSc is the Schmidt number. It is derived from the liquid viscosity, the liquid density, and the diffusivity of the solute into the liquid phase. It is used in empirical calculations for the mass transfer coefficient.
Pbot is the pressure at the bottom of the column.
Ptop is the pressure at the top of the column.
PCO2,bot is the partial pressure of the CO2 at the bottom of the column. It is used to find equilibrium values in conjunction with Henry’s Law.
PCO2,top is the partial pressure of the CO2 at the top of the column. It is used to find equilibrium values in conjunction with Henry’s Law.
S is the cross sectional area of the column. It is used in the mass balance to determine the mass transfer coefficient experimentally. It is also used in calculating the liquid film height (HL).
nG is the gas velocity. It describes the speed at which the gas moves through the column.
VG is the volumetric flow rate of the gas. This is the value that is recorded in the lab and later converted to a mass flow rate using the gas density.
VL is the volumetric liquid flow rate. This is the value that is recorded in the lab and later converted to a mass flow rate using the liquid density.
x*bot is the equilibrium value for the mole fraction of solute in the liquid phase at the bottom of the column. It is used in calculating the log mean driving force.
x*top is the equilibrium value for the mole fraction of solute in the liquid phase at the top of the column. It is used in calculating the log mean driving force.
xbot is the mole fraction of solute in the liquid phase at the bottom of the column. It is used to calculate the log mean driving force.
xtop is the mole fraction of solute in the liquid phase at the top of the column. It is used to calculate the log mean driving force.
ybot is the mole fraction of the solute in the gas phase at the bottom of the column. It is used to calculate percent removal as well as the overall mass transfer coefficient.
ytop is the mole fraction of the solute in the gas phase at the top of the column. It is used to calculate percent removal as well as the overall mass transfer coefficient.
μL is the liquid viscosity. It is used to calculate the Schmidt number for determination of the empirical mass transfer coefficient.