| Quantum Mechanics Solution Manual |
|
© Leon van Dommelen |
|
5.2.1.1 Solution hmola-a
Question:
Verify that the repulsive potential between the electrons is infinitely large when the electrons are at the same position.
Note: You might therefore think that the wave function needs to be zero at the locations in six-dimensional space where 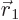

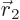 . Some authors refer to that as a
. Some authors refer to that as a Coulomb hole.” But the truth is that in quantum mechanics, electrons are smeared out due to uncertainty. That causes electron 1 to “see electron 2 at all sides
, and vice-versa, and they do therefore not encounter any unusually large potential when the wave function is nonzero at 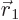

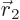 . In general, it is just not worth the trouble for the electrons to stay away from the same position: that would reduce their uncertainty in position, increasing their uncertainty-demanded kinetic energy.
. In general, it is just not worth the trouble for the electrons to stay away from the same position: that would reduce their uncertainty in position, increasing their uncertainty-demanded kinetic energy.
Answer:
The repulsive potential is the term
and when 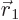

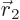 , you are dividing by zero.
, you are dividing by zero.
![]()
![]()
![]() .
.![]()
![]()
![]() .
.